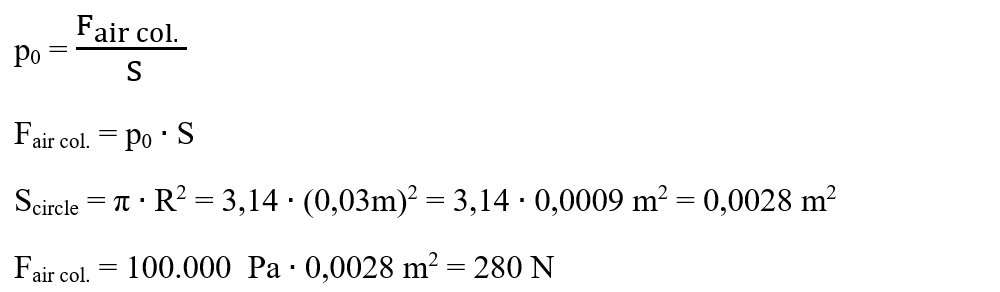V.3. Atmospheric pressure.
Atmosphere is the layer of air that surrounds the Earth.
Until the seventeenth century, it was believed that air was weightless. Galileo Galilei (1564-1642), a great Italian mathematician, physicist and astronomer, first stated that atmospheric air is heavy and that it causes pressure on all bodies on Earth.
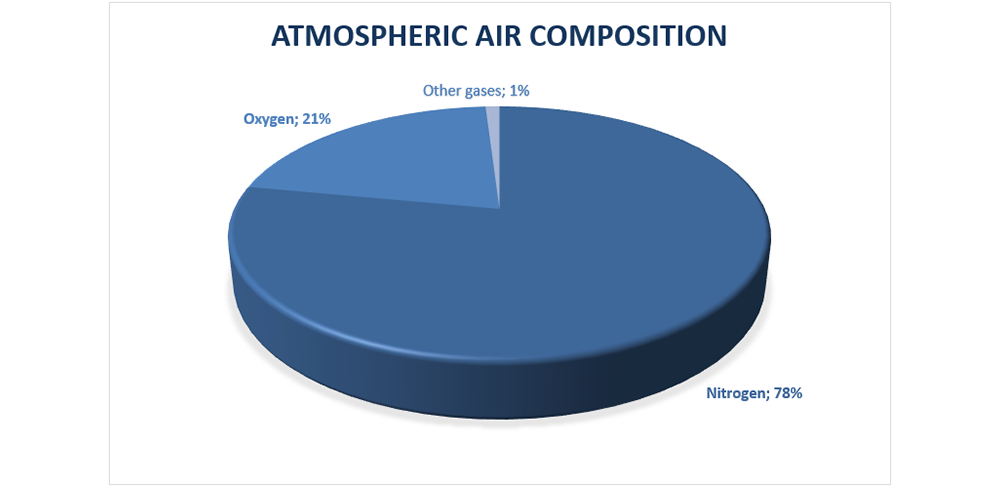
Air composition: 78% nitrogen (N2), 21% oxygen (O2), 1% other gases (argon, carbon dioxide, neon, helium etc.) .

The pressure of atmospheric air on bodies on Earth, due to the weight of the air, is called atmospheric pressure.
Barometers are used to measure atmospheric pressure.
Atmospheric pressure acts in all directions.
👀 Experiment: Side pressure of air on a bag of water.
👀 Experiment: Lateral pressure of atmospheric air on a suction cup.
Required materials:
Suction cup.
Experiment description:
- Press a suction cup to a smooth surface.
- Why is the suction cup stuck to the wall?
When we press the suction cup to stick it to the wall, we remove the air from inside it (it remains empty with pressure 0).
Experiment conclusion:
Due to the lateral atmospheric pressure, the suction cup stays attached to the wall. As long as no air enters it, it is held by air pressure. If the surface of the wall is not smooth and flat, then air remains inside the suction cup, the pressure of which equals the outside and the suction cup falls.
👀 Experiment: Pressing the air from top to bottom on a bag of water.
👀 Experiment: Pressing the air from top to bottom on water from a bottle.
🔥 Caution when working with sharp objects! Be careful not to get stung when working with a needle!
Required materials:
0,5L PET bottle, compass.
Experiment description:
- Take a 0,5L bottle and fill it with water, then put the stopper on it.
- Drill the bottle and you will notice that water does not gush through the hole.
- Unscrew the bottle cap and immediately water gushes through the hole.
When the glass is clogged, the atmospheric pressure does not act on the surface of the water from the bottle.
Experiment conclusion:
When the bottle is uncorked, the atmospheric pressure acts on the surface of the water from top to bottom and water gushes from the bottle.
👀 Experiment: Pressing the air from the bottom up.
Required materials:
Glass of water, cardboard.
Experiment description:
- Cover a glass filled with water with a cardboard so that there is no air between the surface of the water and the paper.
- Push the mouth of the glass over the cardboard and turn the glass upside down.
- Carefully remove the palm from the glass (you can do this experiment above the sink).
Water does not fall from the glass, because the air pressure exerted from the bottom up is higher than the pressure exerted by the water from the glass on the cardboard.
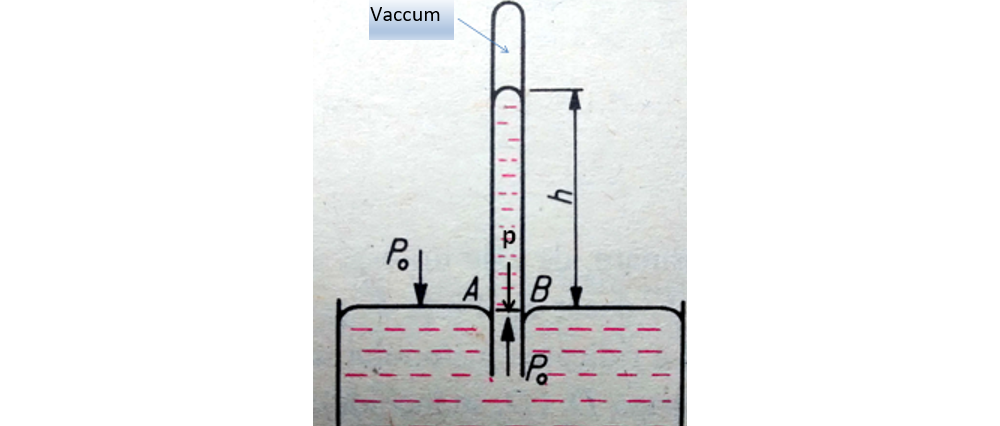
Torricelli's experiment:
The first physicist to determine the value of atmospheric pressure (in 1643) was Evangelista Torricelli (1608-1647), a student of Galileo. Until Galileo Galilei it was known that atmospheric air presses on bodies on Earth, but no one knew its value. Galileo Galilei being near the end of his life asked Torricelli to solve this problem.
Torricelli built the first barometer and measured the value of atmospheric pressure for the first time.
-
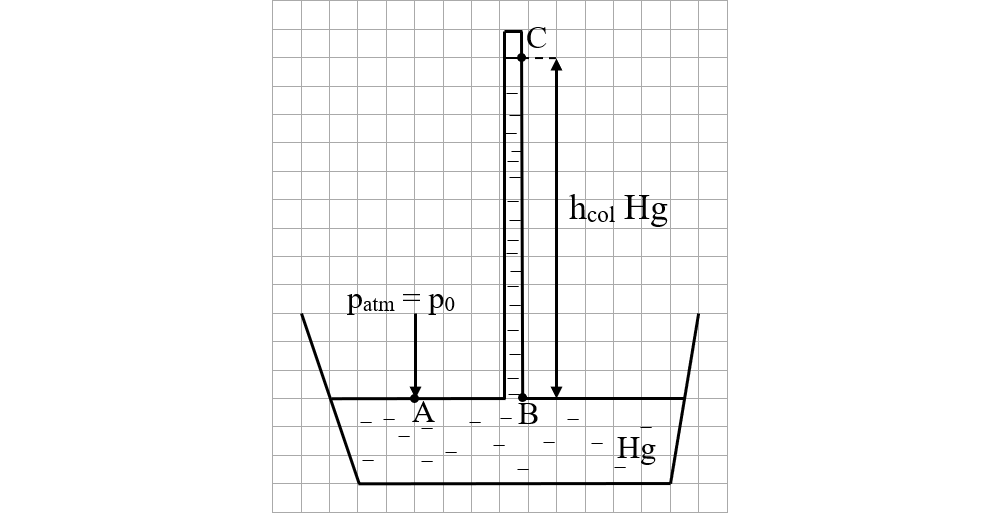
Torricelli filled a one-meter-long glass tube, which was very thin and closed at one end, with mercury, which he overturned in a mercury vessel.
-
He noticed that only a small amount of mercury (Hg) was spilled into the vessel.
-
Measured the height of the mercury column left in the tube, noted
hcolHg = 76 cm = 0,76 m
-
To calculate the atmospheric pressure he applied the formula of the hydrostatic pressure of the mercury column remaining in the tube.
-
At the same level in a liquid the pressure is the same, ie pA = pB.
pA = atmospheric pressure, denoted by p0
pB = the hydrostatic pressure of the mercury column in the tube = ρHg ∙ g ∙ hcolHg
pC = vacuum pressure = 0
p0 = ρHg ∙ g ∙ hcolHg = 13600 ∙ 9,8 ∙ 0,76 = 101.325 Pa
This is the value of atmospheric pressure.
Atmospheric pressure varies all the time and depends on two factors:
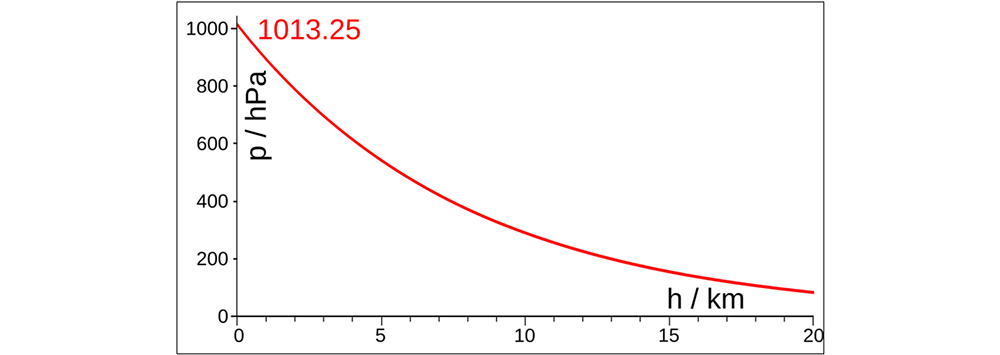
1) The higher the altitude, the lower the atmospheric pressure because the air becomes thinner (ie the concentration of oxygen and nitrogen molecules decreases). At altitude 0 (sea level): p0 = 101.325 Pa.
2) Weather (rain, air temperature, winds etc.).
🔐 Applications
Variation of the atmospheric pressure with altitude (decrease in atmospheric pressure with increase in altitude) was demonstrated by the French physicist Blaise Pascal (1623-1662). He pointed out that the rise of liquids in a vacuum due to atmospheric pressure (ie the height of the liquid column) decreases if the experiment is performed at high altitudes (on a mountain).
At Everest Peak, the highest point on Earth, with an altitude of 8848 m above sea level, the atmospheric pressure is about a third of that at sea level, ie p ~ 33.800 Pa.
Based on the dependence between altitude and atmospheric pressure, altimeters are built, used on board aircraft, which are in fact barometers calibrated directly in meters. They measure altitude (height above the Earth's surface).

Jet aircraft flight
Here is a graph showing the decrease in atmospheric pressure (p) with increasing altitude (h):
Flying at very high altitudes has several advantages:
- flight in the stratosphere (over 12.000 m) guarantees the absence of turbulence, being above atmospheric disturbances
- reducing air density at high altitudes implies a reduction in aircraft traction and a corresponding increase in speed.

However, even the fact that the atmospheric pressure drops rapidly with increasing altitude causes serious inconvenience, especially for crew and passengers: oxygen pressure becomes dangerously low above 8,000 meters, and biological tissues over 12,000 m can suffer very serious damage due to the decrease pressure. Therefore, all jet-powered aircraft (jet engines) must be equipped with pressurization systems.
Modern transport aircraft do not maintain atmospheric pressure in the cabin at sea level (1.013,25 hPa), but a pressure equivalent to that of an altitude of approximately 2.500 m. The variation of the cabin altitude during the ascent and descent of the aircraft is maintained by the pressurization control system. In essence, the aircraft uses some of the excess air that is drawn by the compressors into its jet engines. Engines do not need all the air for combustion, so some of it is extracted and used for both air conditioning and pressurization. Excess air from the compressors is cooled and then pumped into the cab.
How much air is needed to apply pressure depends on the volume of the cab. Because the aircraft's pressurization system works in combination with the air conditioning system, it continuously circulates air through the cabin, recirculating some of it and venting the rest, as it draws in fresh air from the engine compressor.
The air pressurization system also contains safety mechanisms designed to prevent accidents. The positive pressure relief valve will open if the pressure inside becomes too high because too much air is being pumped into the cab. It will relieve that pressure. There is also a negative pressure valve, which protects the aircraft from the effects of a change in which the outside pressure would become higher than inside the cabin during a sudden descent.
Depressurization may occur due to failure of the compression system, due to human or technical errors, due to severe turbulence, cracks in the windshield / window / fuselage, doors that do not close tightly etc.
1. Explosive decompression: occurs at a fast pace, usually in 0,1 - 0,5 seconds, being caused by breaking a window, opening a door, a hole in the fuselage. The risk of pulmonary trauma is very high. What happens in case of rapid depressurization:
- oxygen masks fall off automatically. The oxygen mask provides oxygen to the passengers, while the pilots lower the aircraft to the safety ceiling of 2400 meters (8000 ft). Oxygen is not unlimited. It was calculated for a few minutes (up to 15 minutes), the time required for the plane to reach an altitude of 2400 meters where the air is breathable.
- there is a loud noise and the noise level increases in the cabin
- the cabin is covered with condensation, like a fog
- objects start flying in the cabin, uncontrolled
- the water starts to boil instantly.
2. Slow / gradual decompression: takes a long time, up to a few minutes. This type of depressurization can only be detected with on-board instruments. If left untreated, slow uncontrolled depressurization can lead to hypoxia. Slow depressurization can occur when a door has not been sealed or when there are problems with the pressurization system. Passengers' ears are clogged, abdominal pain or hearing discomfort may occur, noise levels increase in the plane.
The biggest danger in case of depressurization is hypoxia (insufficient oxygen supply in the body), as well as the possibility of being pulled from the plane (in case of explosive depressurization).
Variation of atmospheric pressure depending on the weather
Air flows from high pressure areas (called anticyclones) to low pressure areas (called cyclones). Although the air is colder and wetter in high pressure areas, meteorologists predict good weather with clear skies as the wind pushes the clouds into the surrounding areas with lower pressure. So clear skies, no threatening clouds and dry air are predicted by increasing atmospheric pressure. Strong winds are signaled by the situation where high pressure areas are very close to low pressure areas.
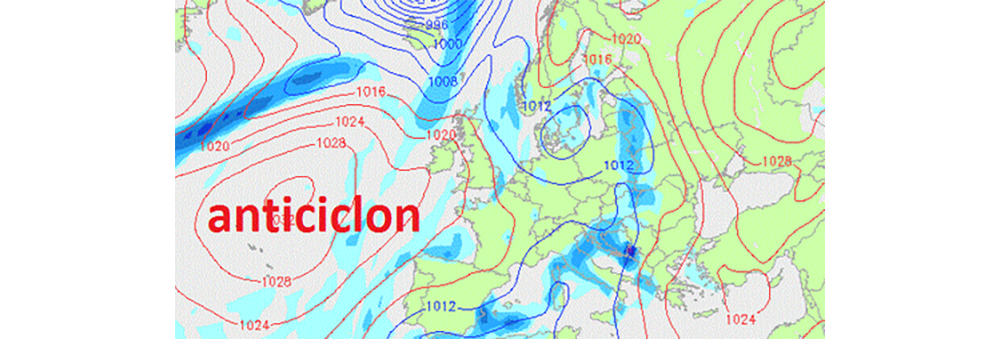
Due to the continuous variation of the atmospheric pressure, it was necessary to choose a reference pressure, called normal atmospheric pressure, denoted by p0.
p0 = 101.325 Pa = 1 atm = 760 mmHg (it is measured at sea level at 0 °C).
Atmospheric pressure applications
Do you feel the pressure of the air? The answer is NO. Why don't we feel the pressure of the air? Our body has an internal pressure (especially given by the fluids in us: water, blood etc.) that equals the external pressure of air.
But what would happen to us if we went out into space, where there is a vacuum and the pressure is zero? At pressure = 0, the water no longer boils at 100 °C, but at 0 °C and in a few seconds our blood would start to boil and we would die. That's why cosmonauts when they go into outer space use special cosmonaut suits that are pressurized (they have water tubes inside) which replace air pressure.

On planet Venus, the surface temperature reaches up to 450 degrees Celsius, and the atmospheric pressure is huge, 92 times higher than that of Earth. There the pressure of the atmosphere (composed of 96.5% carbon dioxide, 3.5% nitrogen) would simply crush us.
A famous experiment to highlight atmospheric pressure was carried out in 1650 by Otto von Guericke, who used two hemispheres sealed together and vacuumed inside. People tried to detach the hemispheres on which the atmospheric air pressed, but they failed. Eight horses on each side were harnessed to separate the two hemispheres. The hemispheres can still be found today at the German Museum in Munich.

👀 Experiment: Gas law and how to breathe.
Required materials:
Vessel with a perforated tube stopper, 3 Berzelius glasses, colored water, hot water and cold water.
Experiment description:
- Take a bowl with a perforated stopper with a tube.
- Insert the tube into another bowl of water.
- Heat the air in the tube vessel. What do you notice?
When we heat the air in the tube, the air enters the water. As the temperature rises, the air expands, increases its volume and decreases its pressure compared to the atmospheric pressure (outside).
- Cool the air in the tube. What do you notice?
When we cool the air in the tube, water rises into the tube. When the temperature drops, the air contracts, decreases its volume and increases its pressure compared to the atmospheric pressure (external) one.
Experiment conclusion:
The gas pressure is inversely proportional to their volume, ie as the pressure increases, the gas volume decreases and vice versa.
Mathematically we write:

We have seen that the volume of the gas increases in direct proportion to its temperature.
Mathematically we write:

By combining the two equations, we obtain the law of ideal gases:

Remark:
At constant volume the pressure is directly proportional to the temperature (this is not the case in this experiment, because the volume of the air has either increased or decreased).
Application of the Gas Law
One application of the inverse proportionality of pressure to air volume is human respiration.
So:
-
On inspiration, the volume of the lungs increases, the internal pressure decreases compared to the external one (atmospheric) and the air enters the lungs.
-
On expiration, the volume of the lungs decreases, the internal pressure increases compared to the external one (atmospheric) and the air leaves the lungs.
🔓 Solved problems
1. Calculate the compressive force that the atmospheric air exerts on a window with a length of 1 m and a width of 60 cm, knowing that the air pressure is 105 Pa.
Solution:
We write the problem data:
L = 1 m
l = 60 cm = 0,6 m
p = 100.000 Pa
F = ?
We write the pressure formula and remove the unknown, F:

We calculate the window area and replace the data:
S = L ∙ l = 1 m ∙ 0,6 m = 0,6 m2
F = p ∙ S = 100.000 Pa ∙ 0,6 m2 = 60.000 N
We notice an enormous force from the air on the glass and yet the glass does not break, because the air acts both externally and internally with the same force, having a resulting force equal to zero.

2. How long should Torricelli's tube be if we used water instead of mercury? The normal atmospheric pressure is 100.000 Pa and the water density is 1000 kg/m3.
Solution:
We write the problem data:
hcol. apă = ? m
p = 100.000 Pa
ρ = 1000 kg/m3
We write the atmospheric pressure formula and remove the unknown, hwater col.:
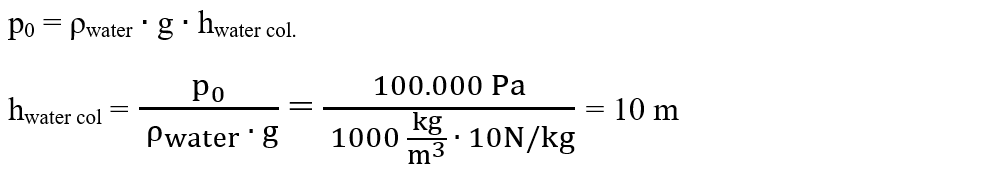
3. A glass of 400 cm3 with a mouth diameter of 6 cm, filled with water, is covered with a cardboard and overturned. The density of the water is 1000 kg/m3.

Is required:
a) What force does water exert on the cardboard?
b) What is the compressive force exerted by the atmospheric air on the cardboard, knowing the air pressure of 100.000 Pa?
Solution:
We write down the problem data and turn it into SI:
Fwater col. = ?
Fair col. = ?
p = 100.000 Pa
ρ = 1000 kg/m3
V = 400 cm3 = 400 ∙ (0,01m)3 = 0,0004 m3
D = 6 cm
Rcircle = D/2 = 3 cm = 0,03 m
The force of the water is the weight of the water in the glass:
Fwater col. = Gwater col. = m ∙ g = ρwater ∙ V ∙ g = 1000 kg/m3 ∙ 0,0004 m3 ∙ 10 N/kg = 4 N
The force of the air is found from the pressure formula: